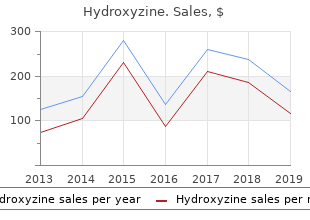
Hydroxyzine
"Cheap hydroxyzine 25mg fast delivery, anxiety reddit."
By: William A. Weiss, MD, PhD
- Professor, Neurology UCSF Weill Institute for Neurosciences, University of California, San Francisco, San Francisco, CA

https://profiles.ucsf.edu/william.weiss
Eforts to pilot the Government and industrys current preferred option of an upfront payment scheme should not be delayed cheap hydroxyzine 10mg without a prescription anxiety symptoms not going away. We recommend that other options to address market failure 10 mg hydroxyzine free shipping anxiety symptoms on kids, including changes to patent law for antimicrobials and Lord ONeills play or pay proposals 25mg hydroxyzine visa anxiety and high blood pressure, should also be considered by Government buy hydroxyzine 25 mg cheap anxiety 4th 9904. A transferable patent voucher would grant a patent extension to the successful developer of specifed antimicrobials; this patent extension could be used to extend the patent on another product, or could be sold on to another company. We also heard that progress was also being made in reducing antibiotic prescriptions. In primary care, there has been a 13% reduction in prescriptions in the past fve years. However, from 2015 to 2016 hospitals reduced their use of the ultra-broad spectrum antibiotics piperacillin/tazobactam and carbapenems (both -4%). This is the frst step in reducing antibiotic use in hospitals and focussing on using these antibiotics appropriately is key to preventing the emergence and spread of carbapenem-resistant Gram-negative bacteria. We heard that in secondary care much lower target reductions, of around 1–2%, have been set. This is because many people requiring hospital admission are acutely ill and are prescribed an antibiotic on admission as an appropriate safety measure while tests are run. Many decisions to prescribe antimicrobial medicines are made in the absence of a positive diagnosis of the cause of the infection, potentially leading to inappropriate prescriptions. For example, as we note above, although only 10 per cent of sore throats beneft from antibiotic treatment as most are viral, antibiotics are still prescribed in as many as 60 per cent of cases. Lord ONeill argued strongly for the increased availability of rapid diagnostic testing, and, ultimately, for all antimicrobial prescribing to be tied to diagnostic testing. This is because, even if a patient tests positive for a certain bacteria, that does not mean that antibiotics are required, as in many cases the illness will be self-limiting and the persons own immune system will manage to fght it of without antimicrobial treatment. We were told that decision-making algorithms could in some cases be as efective or more efective than diagnostic tests in improving prescribing, and far cheaper. Professor Michael Moore, Professor of Primary Health Care Research at the University of Southampton, told us: The research is published, but it just does not get into practice. You are saying why isnt there a thing coming up on my screen to say is it an antibiotic? If you say to a general practice, which is a small business, “We want you to use this test, they say that it costs £10 a go, whereas it costs them nothing not to use it. If they are doing 49 Q140 50 Qq13–14 51 Q90 52 Q91 53 Q91 16 Antimicrobial resistance 100 of those tests a year, you are asking them to spend £1,000 on that testing. It is about getting the resource in the right place to get these things implemented. Digital health tools for clinicians and policymakers have the potential to greatly increase the quality, safety, and cost efectiveness of clinical care and reduce the threat of antimicrobial resistance. DataLab argue that whilst sofware exists that supports prescribing, its use to support antimicrobial and other prescribing is variable, and that better co-ordination is needed at a national level: 54 Q90 55 Q151; Qq153–154 56 openprescribing. Practically this is normally done by generating a pop-up box for the prescriber before they fnally issue a prescription. It appears that there are diverse arrangements and difuse responsibility for procurement and assuring that these systems are high quality, and providing front line health clinicians with support based on the best evidence and latest national guidance. In order to preserve the efectiveness of current antimicrobial medicines for as long as possible, it is essential that they are prescribed appropriately. As prescriptions are now issued electronically, introducing prompts to prescribers which require them to ensure that correct algorithms, diagnostic tests and reviews have been followed before antimicrobials are prescribed should be a simple matter, making appropriate prescription of antibiotics the default option. Digital health tools for clinicians and policymakers have the potential to greatly increase the quality, safety, and cost efectiveness of clinical care and reduce the threat of antimicrobial resistance. We heard a range of views about the importance of rapid diagnostic testing in antimicrobial prescribing, with some arguing that no antibiotic should be prescribed without one, while others felt that this could have unintended consequences, and that in some cases algorithms were a better aid to prescribing. We heard that good progress has been made in reducing antibiotic use in agriculture. Our written evidence expressed persisting concerns about the prophylactic use of antibiotics in animals, and also about the use of antibiotics of critical importance– antibiotics that are defned by the European Medicines Agency as vital in maintaining human health, including fuoroquinolones, 3rd and 4th generation cephalosporins and colistin. We discussed the possibility of changing labelling to inform consumers about antibiotic use standards, and we were told that labelling products as antibiotic-free was in fact misleading as it does not mean that the animal involved had not received antibiotics during the course of its life. Serious concerns remain about the prophylactic or metaphylactic use of antibiotics in animals, and the use of antibiotics of last resort that may as a result lose their efectiveness for humans more quickly. We welcome the Secretary of States clear and strong opposition to any reduction in food standards as a result of Brexit. Antimicrobials can enter the environment through waste–including human, animal and farming waste, as around 70% of antibiotics are excreted in urine. Waste can also come from pharmaceutical companies producing antimicrobial medicines, and is a particular concern in some countries, as our witnesses described: Dr Williams: What is happening? Is a company fushing out the tanks and discharging antibiotics into the environment, and then bacteria breed in that environment? Dame Sally Davies: Some of it is active pharmaceutical ingredients, the preliminary ingredients that are made generally in China and India, and some is the manufacturing. We heard from many of our witnesses that not enough attention has been paid to this area. Sally Davies said that more research was needed,68 a view which was echoed by the Chief Veterinary Ofcer: We need to understand more about how that interaction between the animal, waste, the environment, water and humans drives resistance and what the key pathways are. He added that from 2019, the Environment Agency will be measuring two more commonly used antibiotics, as they will be a better marker than those currently being measured. He told us that afer dairy farmers had been issued with advice not to feed calves with milk from cows who have been treated with antibiotics, they then faced questions on how the milk should be disposed of–there was no scientifc evidence available to guide them on how best to do so. We expect the Governments new strategy to give greater focus and emphasis to this little-understood but important area. The strategy should include commitments to establish safe discharge levels for human waste, agricultural waste and pharmaceutical manufacturing waste, and to introduce systems to monitor and enforce them.
Estimates of the number of infants potentially cloxacillin because suspected bacterial infection [34] generic 10 mg hydroxyzine with visa anxiety symptoms chest pain. Urinary afected by gentamicin toxicity were not undertaken due to alanine aminopeptidase buy hydroxyzine 25 mg with amex anxiety questionnaire for adolescent, urinary β-2-microglobulin purchase hydroxyzine 25 mg on-line anxiety in dogs symptoms, serum insufcient data discount hydroxyzine 10mg otc anxiety 4 hereford. Given wider scale-up of outpatient-based and urea, and β-2-microglobulin were measured during and after lower-level treatment of possible serious bacterial infection, the end of treatment to detect signs of renal toxicity. Levels improved data are essential to better assess the risk from of urinary aminopeptidase increased in 12 infants, indicating neonatal gentamicin treatment without assessment of blood damage to cells of the proximal tubuli. Serum levels of urea and β-2- A Bartter-like syndrome is a toxic manifestation of gentamicin microglobulin did not indicate any drug-associated depression which includes nephrotoxic syndrome. Hypocalcaemia, Ten infants were treated with gentamicin and 10 infants not- hypomagnesaemia, alkalosis, and hypokalemia were the main treated served as controls. After discontinuation of rate and the fractional excretion of β-2-microglobulin in gentamicin, recovery of the tubular functions and resolution urine were used as indicators of renal dysfunction [35]. The fractional excretion of of 7 bacteria are reported in table 2, by Sader, et al. The β-2-microglobulin was statistically higher in 4/10 of infants % susceptibility ranges from 37. However, the observed renal Gentamicin is not metabolized and is excreted unchanged dysfunction is reversible. In literature, there are not studies on the Toxic efects of gentamicin in infants metabolism of gentamicin in infants and children. Potentially toxic trough concentration (> 2 µg/ml) of gentamicin were recorded in 57 Fifty full-term infants (gestational age 37-42 weeks, and birth of 91 (63%) infants; 24 of these concentrations were 3 µg/ml. Toxic trough concentrations were not accompanied by raised peak serum Table 2: Susceptibility to gentamicin and typical minimal values. To prevent accumulation, the dosage interval may need to Acinetobacter baumannii 37. Once- remaining 6 infants with estimated gestational age between daily dosing had the highest peak and the lowest trough 26 and 30 weeks and birth weights between 720 and 1,450 gentamicin concentrations. Capillary (heel prick) blood Gentamicin pharmacokinetics in low-birth weight samples were collected at 0, 0. For both dosage regimens in 41 preterm low-birth weight infants (20 infants with birth plasma gentamicin levels were monitored during a dosage weight of < 1,500 gram and 21 with birth weight of ≥ 1,500 interval on three separate occasions over a 10 day period. Gentamicin regimens, were regimens gave satisfactory plasma gentamicin concentrations 2. The body clearance of gentamicin correlated of treatment, the half-life and the total body clearance were signifcantly with gestational age (r = 0. In a one-compartment these results indicate that either regimen may be useful in the pharmacokinetic analysis, a large variability among infants clinical situation but from a practical standpoint administration was observed on the 1st day of treatment. The gentamicin every 24 hours may be easier to comply with then every pharmacokinetic parameters are shown in table 4. Gentamicin pharmacokinetics in very-low-birth weight Gentamicin pharmacokinetics in small-for-gestational age Very-low-birth weight preterm infants (N = 18) with suspected and appropriate-for-gestational-age infants infection were administered gentamicin intramuscularly every 18 hours (2. Lulic-Botica [43] compared gentamicin pharmacokinetics the gestational age and the body weight ranged from 26 to among infants born small-for-gestational age and appropriate- 32 weeks and 690 to 1,450 gram, respectively. These authors further compared gentamicin infants with estimated gestational age ranging from 26 to pharmacokinetics in subgroups of appropriate-for-gestational 32 weeks and birth weights ranging from 690 to 1,420 gram age and small-for-gestational age infants born preterm and were administered 2. Table 4: Gentamicin pharmacokinetic parameters in low-birth weight infants in the frst week of life. Elimination rate Distribution volume Total body clearance Infants -1 Half-life (hours) (hours ) (L/kg) (L/kg/h) < 1,500 gram group 1st day of treatment (N =19) 0. Statistical analysis included X2 and hours for those who weighed 1,200 – 2,000 gram, and 4 - 5 non-parametric Mann-Whitney-U-test. A total of 1,179 studies were screened and 82 articles were In infants aged > 7 days of life, the only parameter diferent identifed as eligible for inclusion. Approximately Some authors suggested to extend intervals among gentamicin half of all Staphylococcus aureus isolates were methicillin- doses in premature infants when the trough gentamicin resistant. After age stratifcation, the median rate of resistance concentration is > 2 µg/ml to yield a trough gentamicin of common gram-negative bacteria to gentamicin/amikacin concentration < 2 µg/ml. The gentamicin concentrations and was extremely high (Klebsiella pneumoniae 75%, Escherichia intervals among gentamicin doses are summarized in table 8 coli/gentamicin 55. Culture positivity Research is lacking regarding the treatment of suspected rate was found to be 15. Most common bacterial isolate was sepsis in infants and children with hospital-acquired sepsis, Escherichia coli (45. Maximum gram- and penicillin for hospital-based patients or gentamicin negative isolates (50%) showed high resistance to gentamicin intramuscularly or intravenously and oral amoxicillin when and other antibiotics. These authors recommend continuous referral to a hospital is in accordance with currently available monitoring of changes in bacterial pathogens causing urinary and other international guidelines, and there is no strong tract infection and antibiotic sensitivity in each area for efective evidence to change this. Gram- 386 infants with very-low-birth weight, among which 262 negative/gram-positive bacteria was found in 68% to 25. The mortality the most common species was Streptococcus epidermidis rate was 79% caused by multidrug-resistant organisms. Gram- (63%), then Streptococcus haemolyticus (28%) and other positive and gram-negative bacteria showed high resistance central nervous system pathogens (9%). There is an alarming increase in antibiotic resistance and Among Streptococcus haemolyticus isolates, 100% were continuous surveillance for antibiotic susceptibility is needed resistant to gentamicin.
It has been found in Gram-positive and Gram- Transposon negative bacteria order 25mg hydroxyzine with visa anxiety treatment for children, aerobic and anaerobic bacteria hydroxyzine 10 mg mastercard anxiety chest tightness, and in all Plasmid environmental and biological niches94 buy hydroxyzine 10mg cheap anxiety jaw pain. The pneumococci have shown that the chromosomal location of the resistance determinant is not a safeguard against its spread discount 10 mg hydroxyzine anxiety symptoms stomach pain. Bacteria themselves are mobile and can easily travel from person to person and between countries. Resistant pneumococci in Iceland and in the United States have been shown to be the progeny of strains that initially appeared in Spain85. Thus, countries and Chromosome citizens worldwide have become part of a global microbial ecology, tn tn sharing and spreading the consequences of antimicrobial resistance. Some are directed at the antibiotic itself: enzymes such as β-lactamases destroy penicillins and cephalosporins, and modifying enzymes inactivate chloramphenicol Antibiotic-resistance Antibiotic- and aminoglycosides such as streptomycin and gentamicin. Others genes efflux pump target how the drug is transported; for example, an active efflux of drug mediates resistance to the tetracyclines, chloramphenicol and the fluoroquinolones98,99. For example, the β-lactamases now number in the hundreds and more than 20 different resistance determinants mediate an efflux of tetracyclines100. In addition, more than one type of mechanism may provide resistance to the same antibiotic; for Chromosome Antibiotic- example, tetracycline resistance can be effected by either efflux or altering Antibiotic ribosome protection101. Although most fluoroquinolone resistance enzyme stems from chromosomal mutations in the gyrase target or from drug efflux, a plasmid-mediated resistance to fluoroquinolones has been recently described102. Multidrug resistance can be specified by chromosomal genes for regulatory proteins such as MarA and SoxS. These proteins promote drug resistance by controlling the expression of other chromosomal genes, Bacterial cell such as those involved in drug efflux61. Ecologically speaking, it is the density of antibiotic usage that Some studies have, however, tracked a decline in resistance frequen- enhances resistance selection and its effects. Asignificant countrywide reversal involves the total amount of antibiotic being applied to a geographi- of macrolide resistance in S. Nonetheless, resistance generally becomes a factory of resistant bacteria that enter the environment. The end result of the selective pressure will reflect the number to a decrease in resistant strains. For example, despite being put of individuals who are contributing resistant bacteria to that environ- into clean cages, chickens previously fed tetracycline-laced feed ment and the residual number of surviving, susceptible bacteria. They are societal drugs in which individual use affects others new location in the barn every 2–3 days, however, the resistance fre- sharing that environment62,63. In Nepal, resistance rates in individuals were found to correlate more with the total community use of antibiotics the ecology of antibiotic resistance than with the individuals own use66. The impact of the drug selection process can be largely confined to In addition,the selection of resistance continues because antimicro- the individual taking the antibiotic if widespread antibiotic usage is bials persist, largely intact, in natural environments. After therapy, the selected resistant commensal strains will waste waters are being reported with increasing frequency and are eventually be diluted outand their growth will be suppressed by the potentially important contributors to the environmental selection of return of drug-susceptible, natural competitors. The findings suggest that one populations are being treated with the same class of antibiotic, sus- approach to the antibiotic resistance problem could be to design drugs ceptible strains will have little opportunity to recolonize their niche that self-destruct after treatment,thereby removing a contributing fac- and resistant strains will acquire an important advantage. Confronted with a shortage of Considerable debate surrounds the relationship between antimicro- new antimicrobials, we must use our current drugs more prudently. The Reducing and improving use can diminish resistance and permit a chronic use of subtherapeutic amounts of antibiotics for growth drug to resurface eventually as an effective therapy58. The appropriate promotion in food animals has been banned in the European Union, use of the antibiotics not only can help to reverse high resistance fre- but it continues in the United States, albeit under intense scrutiny by quencies, but also can curb the appearance of resistance to newer the Center for Veterinary Medicine of the Food and Drug agents58. Despite their low-level application, the antibiotics hospital units has shown that susceptible indigenous strains will select determinants mediating high-level, clinically relevant resist- repopulate the ecological niche in the absence of drug-selective pres- ance55. In addition, such efforts cannot succeed may occur through the food chain or through animal handlers68–71. Continued use of the same drugs in infections are small but not insignificant; they have a major role if areas where resistance is endemic should be halted. These varied applications all add to cumvent resistance mechanisms or those that attack new targets—is the continued selection of resistant bacteria. Such antibiotics would evade current resistance mecha- nisms, which can thwart the success of new, but structurally similar How can we manage and prevent drug resistance? A different approach focuses on preventing infection by inhibit- Track the resistance frequency. Local, national and global surveillance ing key gene products that are involved in the infection process itself80. Such surveillance would alert public health officials to new pathogens and would spur the implementation of control poli- cies. In this regard, the Alliance for the Prudent Use of Antibiotics has a established its Global Advisory on Antibiotic Resistance Data project to synthesize, evaluate and report the surveillance data from five large global surveillance systems (Box 2). Commensal organisms are common reservoirs of antibiotic resistance plasmids, transposons and genes. The commensal Haemophilus parain- fluenzae has been shown to confer β-lactamase-specifying plasmids to H. Similarly, Staphylococcus epidermidis serves as a reservoir for resistance genes and plasmids for the more pathogenic S. Vancomycin resistance determinants found initially c among enterococci appeared in other commensal bacteria before emerging in S. This concept has been recently formalized by an Alliance for the Prudent Use of Antibiotics–based Reservoirs of Antibiotic Resistance project that supports studies examining the link between resistance in commensal flora and resistance in clinical isolates (Box 2). Isolate hospitalized individuals with potentially dangerous resistant bacteria: cohorting.
To a large extent the process outlined in the long-term supply continuity model was followed cheap hydroxyzine 10 mg without a prescription anxiety symptoms 3 months. But the assessment of partial and fully delinked models is important evidence that should be taken into account when considering market entry rewards order 10 mg hydroxyzine anxiety symptoms postpartum. The stakeholder project group perceived little value in implementing a fully delinked model buy cheap hydroxyzine 10 mg on-line anxiety lost night, given that antibiotics are generally already used responsibly and sparingly in Norway cheap 10mg hydroxyzine mastercard anxiety symptoms checklist. The power of the pharmaceutical industry to over-promote an antibiotic in Norway is considered marginal, and the cost of implementing a dedicated delinked system for only a handful of medicines was considered too bureaucratic and costly. The payer then takes full responsibility for production, supply, distribution, pharmacovigilance and additional regulatory applications to extend the geographic availability of the antibiotic. The developer will expect to be paid the full value of the antibiotic at registration, including the development costs. Yet if it is perceived as too difficult to achieve, it may result in more companies exiting antibacterial R&D. Determining which types of antibiotics are eligible for a market entry reward is a central design decision. However, under existing market conditions of limited prices and highly restricted use, achieving this level of sales is unlikely for a new antibiotic reserved for treating only patients with specific multi-drug-resistant infections. For example, with the existing pipeline it would apply to about two antibiotics within the next five years. If this definition is applied historically, based on unmet public health threats at the time, potentially only three antibioticsxi (not used solely for tuberculosis) would have received this award in the last twenty years. A broadly inclusive reward would incentivize incremental innovation, potentially leading to therapeutic improvements that would not be developed if only the first-in-class antibiotic was rewarded. As a result of the expanded eligibility profile, a broadly inclusive reward would be triggered relatively more frequently. For example, with the existing pipeline it would apply to about ten antibiotics within the next five years. The simulation indicates that without any intervention 14 known-class antibiotics (range of four to 26) offering therapeutic improvements would receive marketing authorization during the next 30 years. Of course, the two types can also be combined where the value of the market entry reward varies depending on the characteristics of the antibiotic. For example, bigger rewards could be given for antibiotics meeting the tightly focused criteria, and smaller ones for the broadly inclusive. This would encourage risk-taking to pursue new classes but still incentivize diversity and improvements to existing classes. A limit should be placed on the number of known-class antibiotics incentivized, to avoid a proliferation of similar products. Table 4: Strengths and weaknesses of a tightly focused vs broadly inclusive market entry reward Type Strengths Weaknesses Creates clarity via a focus on Owing to high discovery challenges and innovation development risk with low probability of success, this may be non-incentivizing, leading May drive R&D investment to take to divestment and exit from this therapy area new risks by incentivizing innovation across all phases of R&D Tightly focused Increases the diversity of antibiotics to A high bar for innovation increases the risk of slow the selection of resistance failure, which may discourage private capital investment in antibiotics in favour of other Focuses public investment on therapeutic areas innovation and the highest public xi Linezolid, daptomycin and fidaxomicin. It provides an opportunity for payers to engage formally with industry to facilitate the development of novel antibiotics and better understand the future budget requirements for the antibiotic pipeline. Prequalification would also provide an opportunity for payers and developers to discuss sustainable use and equitable availability criteria. Prequalification may also encourage more private capital investments to help push an antibiotic through the expensive late clinical trials, as it would indicate that a level of due diligence had been undertaken, leading to a positive external assessment of the antibiotic. Moreover, a prequalification process would provide for broad dissemination of knowledge about technical progress in the field. A tightly focused market entry reward of $1 billion (850) per antibiotic (in addition to unit sales revenues) has the potential to bring 18 (13–23) new antibiotic classes to the market in the next 30 years. Figures 12 and 13 show the results of the simulations for different reward categories. These show that the number of new antibiotic classes would more than quadruple to about 18 (range 13–23) by the introduction of a tightly-focused reward scheme with total payouts of $1 billion (850 million) for a partially delinked reward, or $1. The number of new classes plateaus at about 20 new approvals (range 13– 24) in 30 years, if rewards increase to $1. For a broadly inclusive reward, new approvals would more than triple to 55 known-class antibiotics with therapeutic improvements (range 41–73) and with a partially delinked reward of $1 billion, or a fully delinked reward of $1. A plateau of 62 new known-class approvals (range 47–78) is reached if rewards increase respectively to $1. This increase in rewards provides up to about four new classes in both partially and fully delinked models. Other mechanisms (such as a pipeline coordinator) may be more cost- effective for bringing these types of products to market. Figure 12: Market approvals by antibiotic types obtained with partially delinked rewards Note: Antibiotics are placed into two categories depending on the scientific difficulty: “truly innovative and “incremental innovation. In order to simulate the market entry of antibiotics some simplifications were needed regarding the actual complicated process of antibiotic innovation. This particularly relates to the ability to discover and develop new classes or other truly innovative antibiotics. For the simulation, it was assumed that “truly innovative antibiotics enter preclinical development at a rate of 0. Market entry rewards must be bound by sustainable use and equitable availability obligations on the developer. To extend the effectiveness of new antibiotics, in exchange for receiving a reward a developer must accept a set of conditions defined by the payer, including sustainable use and equitable availability and supply (see sections below). These conditions should be detailed in the reward agreement between the payer and the developer.
Order hydroxyzine 10mg with amex. Merkules - ''This Again'' (Official Video).