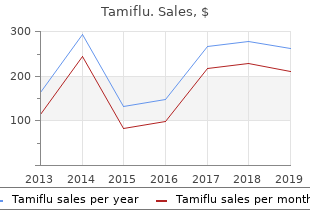
Tamiflu
"Trusted 75 mg tamiflu, 7 medications that can cause incontinence."
By: Richa Agarwal, MD
- Instructor in the Department of Medicine

https://medicine.duke.edu/faculty/richa-agarwal-md
Maculopapular rash induced by diltiazem: allergological investigations in four patients and cross-reactions between calcium channel blockers discount tamiflu 75mg with amex. The spectrum of cutaneous reactions associated with diltiazem: three cases and a review of the literature buy discount tamiflu 75 mg line. Drug Allergy chapter v � 239 Clonidine An adrenergic alpha-receptor agonist used in the treatment of essential hypertension cheap tamiflu 75mg with mastercard. S Incidence Allergic contact dermatitis in 14-38% after transdermal clonidine patch order 75mg tamiflu amex. S Clinical manifestations � Cutaneous: xerostomia (frequent), rash, pruritus, urticaria, angioedema (less frequent), Raynaud�s phenomen (rare), psoriasis exacerbation (rare), lupus erythematosus (rare). Transdermal clonidine patch: local reaction, allergic contac dermatitis, pseudo lymphoma. S Diagnostic methods Skin tests Patch tests (transdermal preparation containing clonidine): positive in a patient who had reacted to transdermal, oral and intravenous clonidine. S Clinical manifestations � General: fever, myolysis, anaphylactic shock (one case with bezafibrate and with bezalip). S Mechanisms Cross-reactive photoallergy to ketoprofen and fenofibrate can be explained by the common ben zoly-ketone structure of these compounds. An association between systemic photosensitivity to fenofibrate and photocontact sensitivity to ketoprofen, tiaprofenic acid, benzophenone and oxybenzone seems to exist. Results of a French nationwide survey of cutaneous side effects of ketopro fen gel reported between September 1996 and August 2000 (Article in French). Cutaneous: pruritus, urticaria, periorbital edema, eczematous rash, bullous pemphigoid, photosen sitivity with bullous dermatitis (frequent), lichen-planus pemphigoides, pseudoporphyria cutanea tardiva, vasculitis, purpura, Sweet syndrome, acute generalized exanthematic pustulosis, erythema multiforme, Stevens-Johnson�s syndrome, toxic epidermal necrolysis, linear IgA bullous dermatitis, papuloerythroderma of Ofuji, hyperpigmented macules. Intradermal skin tests (1%): positive for furosemide as well as for chlorothiazide, bumetanide and sulfamethoxazole-trimethoprim (in one patient). Furosemide challenge in patients with heart failure and adverse reactions to sulfa containing diuretics. Drug Allergy chapter v � 243 Hydralazine A vasodilatator used in cardiovascular diseases. S Clinical manifestations � Cutaneous: lupus erythematosus (+++), one-to-two years after initiation (musculo-skeletal com plaints, fever, weight loss); Sweet�s syndrome, rash, pruritus, angioedema, (rare), fixed drug erup tion, vasculitis, contact dermatitis (occupational). Drug-induced Sweet�s syndrome (acute febrile neutrophilic dermatosis) associated with hydralazine. Hydralazine predisposes to acute cutaneous vasculitis following urogra phy with iopamidol. S Clinical manifestations � Cutaneous: photosensitivity (+++), lupus erythematosus, toxic epidermal necrolysis (rare), vascu litis thrombocytopenic purpura, acute generalized exanthematous pustulosis, lichenoid eruption, angioedema, pityriasis-rosea like reaction. Hydrochlorothiazide-associated noncardiogenic pulmonary oedema and shock: a case report and review of the literature. Hydrochlorothiazide-induced angioedema in a patient allergic to sulfonamide antibiotics: evidence from a case report and a review of the literature. Drug Allergy chapter v � 245 Indapamide A non-thiazide chlorobenzamide sulfonamide diuretic used to treat essential hypertension. It is prepared by condensing chloro sulfonamide acid chloride with an indole amine. S Clinical manifestations Drug induced hypersensitivity syndrome 1/ Maculopapular rash developing > 3 weeks after starting. The two main hypotheses are: 1) Stimulation of T cells by the drug leading to reactivation of herpes virus harbored in T cells. The virus-stimulated T cells show subs tantial cross-reactivity with certain drugs; administration of the drugs leads to an expansion of these specific T cells, which continues after cessation of the drug due to the persistance of the viral anti gens. Association of human herpesvirus 6 reactivation with the flaring and severity of drug-induced hypersensitivity syndrome. Drug-induced hypersensitivity syndrome due to mexiletine hydrochloride associated with reactivation of human herpesvirus 7. Mexiletine-induced severe skin eruption, fever, eosinophilia, atypical lymphocytosis, and liver dysfunction. Drug Allergy chapter v � 249 Pentoxyfylline A purine derivative used as vasodilatator to relieve symptoms in some cases of intermittent claudication. S Clinical manifestations � Cutaneous: generalized exanthematous pustulosis, pruritus, rash, urticaria, angioedema, serum sickness (one case in association with cefoxitin) S Diagnostic methods Skin tests Prick tests negative. S Clinical manifestations � General: anaphylactic shock � Cutaneous: urticaria, angioedema, pruritus, peripheral edema. S Diagnostic methods Skin tests Prick tests are very hazardous (one collapse reported). Prazosin in hypertensive patients with chronic bronchitis and asthma: a brief report. Drug Allergy chapter v � 251 Propafenone A membrane-stabilizing antiarythmic agent resembling propanolol structurally. S Clinical manifestations � Cutaneous: rash, urticaria, angioedema, acute generalized exanthematous pustulosis, psoriasis aggravation, lupus erythematosus-like syndrome. S Risk factors Single-dose intravenous protamine results in protamine-specific IgE or IgG antibody production in 28% of patients. Seroconversion is associated with male gender and insulin-dependent mellitus; these patients may be at increased risk on subsequent exposure. S Diagnostic methods Skin tests (controversial) Intradermal skin tests with 1 �g/ml and 10 �g/ml protamine give false positive results. S Mechanisms IgE or IgG-mediated hypersensitivity (with or without complement activation, unrelated to rate of administration). Complement activation (by heparin-protamine complexes or by interaction with protamine-antipro Drug Allergy chapter v � 253 tamine IgG antibody complexes leading to generation of C3a, C4a, C5a). Increase of thromboxane A2 and 6 ketoprotaglandin F1 alpha, causing pulmonary arterial pressure elevation.
Because of a high incidence of adverse events and the strong association with several autoimmune-like phenomena generic tamiflu 75 mg otc, including myasthenia order tamiflu 75mg with amex, pemphigus generic tamiflu 75 mg without prescription, and Goodpasture disease generic tamiflu 75mg line, the clinical use is limited. The adverse effects of D-penicillamine in animals are similar to those observed in humans. A study on the effects of D-penicillamine in various strains of mice indicated that D-penicillamine facilitates the induction of autoantibodies in animals with an inherent suscep tibility to autoimmunity (Brik et al. Studies using the popliteal lymph node assay demonstrated that D-penicillamine is capable of inducing an antigen. In rats, particularly Brown Norway rats, D-penicillamine induces a disease characterized by dermatitis, vasculitis, production of antinuclear antibodies, formation of circulating immune com plexes, and IgG deposits along the glomerular basement membrane (Donker et al. Interestingly, low 152 Chemical/Physical Agents and Autoimmunity dose pretreatment of D-penicillamine-treated Brown Norway rats was found to induce complete tolerance to a subsequent pathogenic dose of the drug (Donker et al. The syndrome was preceded by influenza-like symp toms, such as fever, headache, muscle and joint pains, and myalgia, which generally started within 6�17 days after starting zimeldine treatment. The British Department of Health and Social Security reported that 400 out of 100 000 patients displayed similar adverse responses to zimeldine. A number of experiments performed thereafter were supportive for the immune-based etiology of zimeldine-induced adverse effects (Kristofferson & Nilsson, 1989). Three individuals occupationally exposed to zimeldine developed allergy to the compound and showed positive patch and skin prick tests and positive response to zimeldine in the lymphocyte transformation test. These findings indicate that zimeldine may be immunogenic; indeed, zimeldine has been shown to be positive in the popliteal lymph node assay, based on cell numbers and including germinal centre formation and production of IgM and IgG antibodies (Thomas et al. The most common adverse effects associated with gold therapy appeared in skin and mucous membranes (about 15% of all patients) and kidneys (about 5�10%), mostly as proteinuria. It is suggestive, moreover, that progressive interstitial lung fibrosis was found in gold therapy (Smith & Ball, 1980), possibly with an autoimmune pathogenesis. Gold therapy occasionally causes autoimmune haemolytic anaemia (Hunziker, 1978), autoimmune thrombocytopenia (Kotsy et al. Early studies showed that injections of gold thiomalate caused renal lesions, immune complex nephropathy, and proteinuria in Wistar rats (Nagi et al. The histology of these lesions can be characterized as either interstitial nephritis or glomerulonephritis, with specific diagnosis dependent on the presence of specific autoantibodies. Recent works using inbred animals have provided additional information on the pathogenesis of gold-induced renal autoimmunity. These findings indicate that gold compounds appear to cause polyclonal B cell activation to induce a variety of autoantibodies, but detailed mechanisms have not been established. The main difference between classical low molecular weight pharmaceuticals and biopharmaceuticals is that biopharmaceuticals are large molecules that can be recognized directly by the immune system, without the need of metabolism or haptenation. Indeed, it has become clear that nearly all biopharmaceuticals induce anti bodies, although many are of human origin and thus immunolog ically tolerated (Schellekens, 2003). The formed antibodies may have no effect at all or are neutralizing, but occasionally adverse reactions may occur. For instance, erythropoietin has been shown to induce autoimmune anaemia in macaques (Chenuaud et al. Treatments with recombinant therapeutic cytokines occasionally induce autoimmune phenomena. Identified risk factors included the female sex, presence of pre-existing autoimmune thyroiditis, the treated disease. However, the immune effects described for diethylstilbestrol depend largely on the age of the animals at treatment, exposure to diethylstilbestrol, and sex. Short-term exposure of mice to diethylstilbestrol has been described as inducing differential immunological effects, depending upon the dose of hormone and sex (Calemine et al. Aged mice appear especially sensitive to diethylstilbestrol treatment, as highly significant alterations were seen in the thymus and bone marrow of aged 21-month-old mice exposed subacutely to diethyl stilbestrol. Severe thymic hypocellularity develops in treated mice following five consecutive days of intraperitoneal injection with diethylstilbestrol. In the same study, the levels of serum IgG and IgM antibodies to cardiolipin showed age dependent fluctuations but were similar in controls and diethylstil bestrol-treated females; however, the IgG antibodies in diethylstil bestrol-treated females were qualitatively different from those in controls with respect to sensitivity to bovine serum (a source of 2 glycoprotein I), although these antibodies are not associated with autoimmune disease. In contrast to the situation with females, diethylstilbestrol-treated males had higher levels of these antibodies than controls (Forsberg, 2000). A potential role of diethylstilbestrol in autoimmunity is also demonstrated by enhanced autoantibody production both in vitro and in vivo. Plaque forming cells producing autoantibodies specific for bromelain treated red blood cells were significantly increased in mice implanted with diethylstilbestrol. IgM antibody production by B1 cells in vitro was also enhanced by diethylstilbestrol treatment. The authors suggested that diethylstilbestrol modulates autoantibody production by B1 cells and may be an etiologic factor in the development of autoimmune diseases (Yurino et al. A number of studies have also demon strated that perinatal exposure to diethylstilbestrol in mice produces profound thymus atrophy; although a direct cause�effect relationship has not been established, this has the potential to influence negative selection processes and subsequently influence autoimmune dis eases. In a follow-up study, using two different groups of diethylstilbestrol-exposed women and an appro priate control group for each, no differences in the prevalence or serum titre of antibodies to five common viral diseases and six less common ones were observed. However, an increased prevalence was found in diethylstilbestrol-exposed women of a relatively rare immunological hyperreactivity, rheumatic fever, subsequent to microbial infection (strep throat) (Blair et al. In a further study (Blair, 1992), sera of diethylstilbestrol-exposed and non exposed women were examined for the presence of factors associ ated with autoimmune diseases. The study demonstrated that the incidence of high antibody titres to red blood cell antigen was higher in the diethyl stilbestrol-exposed females than in the controls. Blair (1992) concluded that, in general, humans exposed prenatally to diethylstilbestrol do not exhibit severe defects in basic immune function, but their propensity to develop autoimmune disease and other diseases associated with defects in immune regulation is increased. There are relatively few data pertaining to risk of specific autoimmune diseases in relation to in utero diethylstilbestrol expo sure, although some studies suggest an increased rate of respiratory tract infections, other infectious diseases, or allergies (Noller et al. However, in a follow-up study of the children born as part of a randomized clinical trial that had been conducted in the 1950s, there was little difference in the rates of reported symptoms or specific diagnoses of infectious, allergic, or autoimmune conditions in diethylstilbestrol exposed individuals (253 sons and 296 daughters) compared with the controls (241 sons, 246 daughters) (Baird et al.
Peer-reviewed medical literature may appear in scientific 75 mg tamiflu otc, medical generic tamiflu 75 mg online, and pharmaceutical publications in which original manuscripts are published buy discount tamiflu 75 mg line, only after having been critically reviewed for scientific accuracy cheap tamiflu 75mg online, validity, and reliability by unbiased, independent experts prior to publication. In-house publications of entities whose business relates to the manufacture, sale, or distribution of pharmaceutical products are excluded from consideration. In determining whether an off-label use is supported, the contractors will evaluate the evidence in published, peer-reviewed medical literature listed below. When evaluating this literature, they will consider (among other things) the following: � Whether the clinical characteristics of the beneficiary and the cancer are adequately represented in the published evidence � Whether the administered chemotherapy regimen is adequately represented in the published evidence. D will be posted to the Web site annually by March 15 for public notice and comment. Public comments will be accepted for a 30-day period beginning on the day the request is posted on the Web site. Content of Requests For a request to be considered complete, and therefore accepted for review, it must include the following information: � the full name and contact information (including the mailing address, e-mail address, and telephone number) of the requestor. If the requestor is not an individual person, the information shall identify the officer or other representative who is authorized to act for the requestor on all matters related to the request. If the complete compendium is available electronically, it may be submitted electronically in place of hard copy. Broad, nonspecific claims without supporting documentation cannot be efficiently reviewed; therefore, they will not be accepted. The information from an internal or external request for inclusion of a therapy in a compendium are available to the public for a period of not less than 5 years, which includes availability on the compendium�s Web site for a period of not less than 3 years, coincident with the compendium�s publication. This information shall be identified and made timely available in response to a public request for a period of not less than 5 years, which includes availability on the compendium�s Web site for a period of not less than 3 years, coincident with the compendium�s publication. This will provide greater clarity to the scope of the Agency�s review of a given request. Submission of Requests Requests must be in writing and submitted in one of the following two ways (no duplicates please): 1. Allow sufficient time for hard copies to be received prior to the close of the open request period. The safety and effectiveness issues pertain to such factors as chemical stability, purity, strength, bioequivalency, and biovailability. Section 1862(a)(1)(A) of the Act requires that drugs must be reasonable and necessary in order to by covered under Medicare. The Medicare Benefit Policy Manual, Chapter 16, �General Exclusions from Coverage,� �180, instructs carriers to deny coverage of services related to the use of noncovered drugs as well. The carrier does not stop Medicare payment for such a drug unless it is notified that it is appropriate to do so through a subsequent instruction. The statute explicitly provides coverage, for blood clotting factors, drugs used in immunosuppressive therapy, erythropoietin for dialysis patients, certain oral anti-cancer drugs and anti-emetics used in certain situations. Beneficiaries are eligible to receive additional Part B coverage within 18 months after the discharge date for drugs furnished in 1995; within 24 months for drugs furnished in 1996; within 30 months for drugs furnished in 1997; and within 36 months for drugs furnished after 1997. For immunosuppressive drugs furnished on or after December 21, 2000, this time limit for coverage is eliminated. Therefore, antibiotics, hypertensives, and other drugs that are not directly related to rejection are not covered. Coverage is available regardless of whether the drug is administered by the patient or the patient�s caregiver. Review of diet and fluid intake for aberrations as indicated by hyperkalemia and elevated blood pressure secondary to volume overload; 2. Reevaluation of the dialysis prescription taking into account the patient�s increased appetite and red blood cell volume; 5. Training of the patient to identify the signs and symptoms of hypotension and hypertension; and 7. Patient Selection the dialysis facility, or the physician responsible for all dialysis-related services furnished to the patient, must make a comprehensive assessment that includes the following: 1. Preselection Monitoring the patient�s hematocrit (or hemoglobin), serum iron, transferrin saturation, serum ferritin, and blood pressure must be measured. Procrit as Preventive Service the carrier may determine that Procrit is covered for individuals who: 1. The preoperative use of Procrit may be afforded to these individuals when carriers, exercising their discretion, determine that this treatment is reasonable and necessary. In other cases, Procrit is considered a preventive service and therefore not covered. The anti-emetic drug is covered as a necessary means for administration of the antineoplastic chemotherapeutic agents. Oral drugs prescribed for use with the primary drug, which enhance the anti neoplastic effect of the primary drug or permit the patient to tolerate the primary anti neoplastic drug in higher doses for longer periods are not covered. Self-administered anti-emetics to reduce the side effects of nausea and vomiting brought on by the primary drug are not included beyond the administration necessary to achieve drug absorption. Only drugs pursuant to a physician�s order at the time of the chemotherapy treatment qualify for this benefit. The dispensed number of dosage units may not exceed a loading dose administered within two hours of the treatment, plus a supply of additional dosage units not to exceed 48 hours of therapy. It is recognized that a limited number of patients will fail on oral anti-emetic drugs. Intravenous anti-emetics may be covered (subject to the rules of medical necessity) when furnished to patients who fail on oral anti-emetic therapy. More than one oral anti emetic drug may be prescribed and may be covered for concurrent use if needed to fully replace the intravenous drugs that otherwise would be given. Hemophilia, a blood disorder characterized by prolonged coagulation time, is caused by deficiency of a factor in plasma necessary for blood to clot.
Obesity: � People with muscular dystrophy often are overweight due to order 75 mg tamiflu with mastercard lack of physical activity buy discount tamiflu 75mg. Contractures: � Contractures are shortening and tightening of the muscle tissue due to generic tamiflu 75mg visa which there is restriction in joint movements cheap tamiflu 75 mg with mastercard. Pressure Sores � Staying for long time in any particular position in muscular dystrophy could result in pressure or bed sore. Food or drink may go down the wrong way (goes to breathing path instead of going to stomach). Complications due to prolonged steroid treatment include: � Cataracts � Cushingoid features(moon face) � Obesity � Short stature � Constipation � Hypertension � Delayed puberty � Behavioral changes (irritability, hyperactivity) � Occasionally slight increase in body hair. May present with pes cavus, unusual hypertrophy (thenar eminence), or patellar subluxation secondary to quadriceps weakness. Autosomal recessive (many sporadic); consanguineous mating increases incidence (for. Popeye arms muscles above elbow are atrophied, those below normal, Strong brachioradialis, but sometimes marked atrophy of biceps. Can be confused with Becker�s Dystrophy,KugelbegWelanderdisease,metabolicmyopathies,congenital myopathies,Polymyositis and acid maltase deficiency Facioscapulohumeral muscular dystrophy (landouzy-dejerine disease). But Childhood form is a more malign disease which runs a more rapidly disabling course. Distal leg weakness of tibialis anterior and toe extensors causes foot drop and slapping gait. Occasionally cauda equina syndrome with leg paresthesias secondary to sway back may occur Involvement usually asymmetrical in distribution and degree. The syndrome of Facioscapulohumeral muscle weakness & wasting can be seen in such diverse conditions as myotubular myopathy, central core disease, nemaline myopathy, myasthenia gravis, polymyositis, adult acid maltase deficiency, & spinal muscular dystrophy. Usually autosomal dominant, onset in third to fourth decade, disease is Progressive with frequently asymmetrical distribution; women affected more than men. Morphologically specific myopathies (So called as benign or congenital myopathies) Major structural types�histological diagnosis. Occasionally rapid progression of disease 445 e) Can overlap with some of metabolic myopathies. Congenital dislocation of hip, particularly in central core disease, Responds poorly to closed reduction because of muscle weakness, usually requires operative stabilization ii. Biopsy reveals specific identifying histological abnormalities as seen by light or electron microscopy & in many cases type l fibre predominance. Metabolic myopathies Myopathies of varying degree, secondary to biochemical defects of muscle metabolism. Glycogenosis the seven recognized type muscles disease associated with enzyme deficiency in the carbohydrate metabolism of muscles are outlined in table. The site of action (liver, heart, muscle, etc) of the deficient enzyme determines the format of the disease. Myopathies are characterised by neutral lipid accumulation in muscle due to mitochondrial metabolic defects. Carnitine (synthesized in the liver & transported via the blood stream) facilitates the passage of free fatty acids 447 across the mitochondrial membrane. This different form may occur at different times in the same patient (biphasic periodic paralysis). Episodic limb weakness usually occurring after exercise, typically spares muscles of respiration & disappear in an unpredictable manner. Lab diagnosis 448 Provocative testing with potassium may be helpful in making the diagnosis. Hypokalemic variety can often be induced by glucose & insulin, heavy exercise followed by rest, excessive alcohol, cold, trauma or stress. Glycogenoses Type Enzyme Deficiency Eponymous or Skeletal Clinical Other tissues features affected other names muscles affected l Glucose-6 Von Glerke�s No phosphatase disease ll 1,4-glucosidase(acid Pompe�s disease Yes a) Severe form: Heart, nervous Generalized; system, kidney, maltase) resembles leukocytes infantile spinal muscular atrophy b) Mild form; Heart resembles limb 449 girdle dystrophy, may present as adult lll Amylo 1,6 Limitdextrinosis Yes Infantile Hepatic glucosidase Forbe�s Disease hypotonia Hypoglycaemia (debranching Cori�s disease Mild Weakness enzyme) Ketosis Leucocytes lV 1,4 � glucan: 1,4 Amylopectinosis Usually no Hepatomegaly � glucan 6 muscle Anderson�s Some Cirrhosis glycosyltransferase symptoms disease cases (branching enzyme; Splenomegaly only In some amylo (1,4 � 1,6) wasting or transglucosidase) weakness V Muscle Phosphorylase McArdle�s Yes Exercise None disease intolerance Muscle cramps Fatigue Myoglobinuria Controlled with high glucose intake and avoiding strenuous 450 activity Vl Liver phosphorylase No Vll Phosphofructokinase Tarui�s disease Yes Exercise Erythrocytes intolerance Muscle cramps Fatigue, Myoglobinuria Malignant Hyperthermia this is a syndrome initiated by a hyper metabolic state of skeletal muscle and characterized by rapid and sustained temperature rise during general anesthesia (surgical stress), accompanied by tachycardia, tachypnea, muscular rigidity, fever, muscle necrosis, cyanosis and severe metabolic and respiratory acidosis. Total body consumption of oxygen increases to two to three times 0 0 normal and temperature can rise (as fast as 1 C every five minutes) to as high as 43 C. Usually expressed as an autosomal dominant trait, but inheritance may be multifactorial. It is important to look for a history of similar problems with anesthesia in the family, though attacks may not necessarily occur with the first exposure to general anesthesia. Musculoskeletal abnormalities such as Ptosis, clubfoot, scoliosis, pectus carinatum, and hernia are common. Treatment If Local or regional anaesthesia is not feasible, general anaesthesia can be accomplished with nitrous oxide, narcotics, barbiturates, ketamine or doperidol. Action: excitation � contraction uncoupling by decreasing release of calcium from sarcoplasmic reticulum. Preanasthetic oral dantrolene loading (4 � 8 mg/kg for 2 days with final dose 2 hours before anaesthesia) may avert or lessen the severity of an episode. It can be improved (fatigues) by repetitive activities, but sometimes this increases its severity (myotonia paradoxica). The repetitive activity persists even though the motor nerve is sectioned or the neuro muscular junction is blocked with curare. It is believed to be a membrane defect (Hyper excitability) related to one abnormality of chloride (Myotonia congenita) or calcium (dystrophia Myotonica) conductance.
Discount 75 mg tamiflu otc. How to open and repair electronic blood pressure/ BP monitoring machine.

