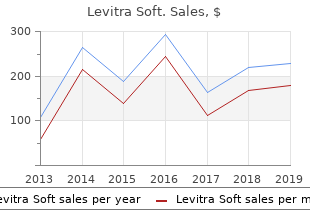
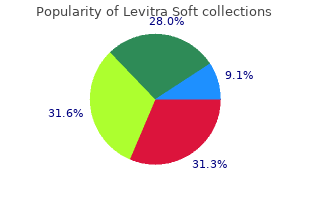
Levitra Soft
"20mg levitra soft for sale, erectile dysfunction fast treatment."
By: Richa Agarwal, MD
- Instructor in the Department of Medicine

https://medicine.duke.edu/faculty/richa-agarwal-md
Upon improvement to safe 20mg levitra soft diabetes and erectile dysfunction causes Grade 1 or less order 20mg levitra soft with visa diabetes-induced erectile dysfunction epidemiology pathophysiology and management, initiate corticosteroid taper and continue to levitra soft 20 mg without prescription erectile dysfunction what age taper over at least 1 month generic levitra soft 20mg visa erectile dysfunction pump youtube. Interrupt or slow the rate of infusion in patients with mild or moderate infusion reactions [see Dosage and Administration (2. A steroid-requiring febrile syndrome, without an identified infectious cause, was reported in six patients (35%) within the first 6 weeks post-transplantation, with five patients responding to steroids. Two cases of encephalitis were reported: one case of Grade 3 lymphocytic encephalitis without an identified infectious cause, which occurred and resolved on steroids, and one case of Grade 3 suspected viral encephalitis which was resolved with antiviral treatment. In animal reproduction studies, administration of nivolumab to cynomolgus monkeys from the onset of organogenesis through delivery resulted in increased abortion and premature infant death. The trial excluded patients with autoimmune disease and patients requiring chronic systemic treatment with corticosteroids (>10 mg daily prednisone equivalent) or other immunosuppressive medications. The most common adverse reactions (reported in at least 20% of patients and at a higher incidence than in the dacarbazine arm) were fatigue, musculoskeletal pain, rash, and pruritus. The most common immune-mediated adverse reactions were rash (16%), diarrhea/colitis (6%), and hepatitis (3%). Across both trials, the most common adverse reactions (reported in at least 20% of patients) were fatigue, musculoskeletal pain, cough, dyspnea, and decreased appetite. The most frequent serious adverse reactions reported in at least 2% of patients were acute kidney injury, pleural effusion, pneumonia, diarrhea, and hypercalcemia. Rate of death on treatment or within 30 days of the last dose of study drug was 4. The most common adverse reactions (reported in at least 20% of patients) were fatigue, cough, nausea, rash, dyspnea, diarrhea, constipation, decreased appetite, back pain, and arthralgia. Treatment could continue until disease progression, maximal clinical benefit, or unacceptable toxicity. The most frequent serious adverse reactions reported in at least 1% of patients were pneumonia, infusion-related reaction, pyrexia, colitis or diarrhea, pleural effusion, pneumonitis, and rash. The most common adverse reactions (reported in at least 20%) among all patients were upper respiratory tract infection, fatigue, cough, diarrhea, pyrexia, musculoskeletal pain, rash, nausea, and pruritus. Table 16 summarizes the adverse reactions, excluding laboratory terms, that occurred in at least 10% of patients in the safety population. After an immune mediated adverse reaction, reactions following nivolumab rechallenge were included if they occurred up to 30 days after completing the initial nivolumab course. Twenty eight patients (11%) had new-onset peripheral neuropathy, and 3 of 40 patients had worsening of neuropathy from baseline. The most common (reported in at least 20%) treatment-emergent laboratory events included cytopenias, liver function abnormalities, and elevated lipase. Other common findings (reported in at least 10%) included elevated creatinine, electrolyte abnormalities, and elevated amylase. After an immune-mediated adverse reaction, reactions following nivolumab rechallenge were included if they occurred within 30 days of completing the initial nivolumab course. The most frequent serious adverse reactions reported in at least 2% of patients were urinary tract infection, sepsis, diarrhea, small intestine obstruction, and general physical health deterioration. The most common adverse reactions (reported in at least 20% of patients) were fatigue, musculoskeletal pain, nausea, and decreased appetite. Table 18 summarizes adverse reactions that occurred in greater than 10% of patients. Immune-mediated hepatitis requiring systemic corticosteroids occurred in 8 (5%) patients. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. There was no evidence of altered pharmacokinetic profile or increased incidence of infusion reactions with anti-nivolumab antibody development. Of patients evaluable for the presence of anti-ipilimumab antibodies, the incidence of anti-ipilimumab antibodies ranged from 6. Overall, there was no evidence of increased incidence of infusion reactions or effects on efficacy with anti-nivolumab antibody development. In animal reproduction studies, administration of nivolumab to cynomolgus monkeys from the onset of organogenesis through delivery resulted in increased abortion and premature infant death [see Data]. Human IgG4 is known to cross the placental barrier and nivolumab is an immunoglobulin G4 (IgG4); therefore, nivolumab has the potential to be transmitted from the mother to the developing fetus. The background risk of major birth defects and miscarriage for the indicated population is unknown; however, the background risk in the U. Nivolumab administration resulted in a non-dose-related increase in spontaneous abortion and increased neonatal death. In surviving infants (18 of 32 compared to 11 of 16 vehicle-exposed infants) of cynomolgus monkeys treated with nivolumab, there were no apparent malformations and no effects on neurobehavioral, immunological, or clinical pathology parameters throughout the 6-month postnatal period. The recommended dose in pediatric patients 12 years of age or greater for this indication is the same as that in adults for this indication [see Dosage and Administration (2. No overall differences in safety or effectiveness were reported between elderly patients and younger patients. In elderly patients with intermediate or poor risk, no overall difference in effectiveness was reported. Nivolumab is an IgG4 kappa immunoglobulin that has a calculated molecular mass of 146 kDa.
Overall survival will be measured from the start of post-progression treatment to generic levitra soft 20mg with mastercard erectile dysfunction medications in india the date of death or levitra soft 20 mg without prescription erectile dysfunction treatment for heart patients, otherwise buy levitra soft 20mg with amex erectile dysfunction treatment clinics, the last follow-up date on which the patient was reported alive discount levitra soft 20mg otc erectile dysfunction blood pressure medications side effects. Accrual to the study will continue until the overall projected sample size of 264 is reached. After these 264 participants are enrolled in the trial, advanced-imaging sites will conduct standard imaging on any additional study participants until all 942 trial participants are enrolled. Assuming 5% attrition of the original sample, we expect that the analysis set will include 250 participants. In particular, in separate univariate models the trans coefficient of change in K (in logarithmic scale) was 0. The following trans table presents computations of statistical power to detect a coefficient for K change of the indicated magnitude, using a two-sided test at level 0. The hazard ratios corresponding to the values of the coefficient considered in the table range from 1. The hazard ratios corresponding to the values of the coefficient considered in the table range from 0. The standard deviation of the predictor was conservatively assumed to vary from 0. Overall survival will be estimated for the entire Advanced Imaging cohort of this sub-study and separately for the two arms using Kaplan Meir curves. The analysis for each of the secondary aims will be carried out similarly to the analysis of the primary aims. For example for secondary aims 1 and 2, Cox regression models will be used with the same predictors as in the primary aims but with progression-free survival as the response. These markers will represent the change between baseline and 22 weeks in measurements derived by imaging. The marker of primary interest will be the volumetric (3-D) tumor measurement on post gadolinium T1 weighted imaging for Aim 1 and the corresponding 3-D tumor measurement on T2-weighted imaging for Aim 2. The primary analytic approach will be based on Cox regression models in which the marker change between baseline and 22 weeks will be the independent variable of interest, with other covariates as indicated in the application. First, Cox regression modeling will be used in which the response variable will be overall survival and the predictors will be measures of tumor size, including 2-D measurements and volumetric (3-D) measurements on post-gadolinium T1 weighted imaging (Kalbfleisch et al, 2008). For each of these measures, the change between baseline and 22 weeks is of primary interest as predictor of overall survival. Models will be fitted for each marker separately as well as for combinations of markers. The c-statistic will be used to assess model fit and cross validation will be employed. The research questions for this Aim are similar to those in Aim 1 but will use T2-based measurements, again with 2-D and volumetric 3-D measurements. However, we do not have information from previous studies on the predictive performance of the markers we plan to explore. Thus, we computed the statistical power to detect a difference in post–week 22 (5. Following the study protocol, we assumed that 612 cases with analyzable data will be available. According to the protocol, approximately 211 deaths are expected in the control arm and 179 in the experimental arm. Assuming now an exponential survival distribution, we calculate that approximately 78% of cases in the control arm and 83% of cases in the experimental arm will survive past 22 weeks. Using the protocol projection of 612 analyzable cases and assuming they will be equally distributed between the arms, we project that about 238 analyzable cases will have survived past 22 weeks on the control arm and about 254 in the experimental arm. The expected number of deaths after the 22 weeks time point would then be 143 in the control arm and 127 in the experimental arm. Using these assumptions we performed power computations for the ability of markers to predict overall survival for participants who survived past 22 weeks. Because we do not have access to any information from previous studies, we performed power computations for two group comparisons, defined by a threshold in the values of the marker. We assumed that the dichotomizing of the marker will generate a “high risk” and a “low risk” group, with the prevalence of the high risk group ranging from 50% (median split on the values of the marker) to 80%. As shown in the table, the available sample size within each arm provides adequate power to detect hazard ratios of 1. Hazard ratios around 2 are considered reasonable for biomarker studies, and so these power calculations suggest our proposal is adequately powered. The occurrence of brain shift after the dura is opened, such as the egress of cerebrospinal fluid, gravity, brain edema, and change in positions of intracranial structures, further complicates optimal surgery (Nabavi et al, 2001). Historically, complete resections have been reported in approximately 20% of cases in surgical series with postoperative imaging (Albert et al, 1994; Barker et al, 1996; Kowalczuk et al, 1997; Simpson et al, 1993; and Vecht et al, 1990). Therefore, the Study Team anticipates that, in the large majority of participants (80% or more), there will be residual disease post surgery that may be measured radiographically. Aims 1 and 2 will examine the ability of the change from baseline to 22 weeks to predict overall survival. When the change is measured as a fraction of the baseline value of the marker, the analysis can be done only with participants that have non-zero values of the marker at baseline. This setting is the primary interest to the proposed study, and so only participants with measurable tumor will be in the main analysis. We present here a second power calculation if there were to be a 20% reduction in our sample size due to this inclusion.
Order levitra soft 20 mg overnight delivery. Curing erection dysfunction ~ Magic cure for erectile dysfunction.
Fractionated proton radiation treatment for pediatric craniopharyngioma: preliminary report cheap levitra soft 20 mg overnight delivery erectile dysfunction implant. Proton therapy for breast cancer after mastectomy: early outcomes of a prospective clinical trial generic levitra soft 20mg with visa erectile dysfunction gluten. Comparison of proton beam radiotherapy and hyper-fractionated accelerated chemoradiotherapy for locally advanced pancreatic cancer buy levitra soft 20mg fast delivery erectile dysfunction doctor karachi. Comparison of adverse effects of proton and x-ray chemoradiotherapy for esophageal cancer using an adaptive dose-volume histogram analysis levitra soft 20mg with amex impotence urinary. Which technique for radiation is most beneficial for patients with locally advanced cervical cancer? Intensity modulated proton therapy versus intensity modulated photon treatment, helical tomotherapy and volumetric arc therapy for primary radiation – an intraindividual comparison. Doses to head and neck normal tissues for early stage Hodgkin lymphoma after involved node radiotherapy. Reirradiation of recurrent and second primary head and neck cancer with proton therapy. Five-year outcomes from 3 prospective trials of image-guided proton therapy for prostate cancer. Comparison of whole-body phantom designs to estimate organ equivalent neutron doses for secondary cancer risk assessment in proton therapy. Proton therapy with concomitant capecitabine for pancreatic and ampullary cancers is associated with a lower incidence of gastrointestinal toxicity. One hundred patients irradiated by a 3D conformal technique combining photon and proton beams. Pencil-beam scanning proton therapy for anal cancer: a dosimetric comparison with intensity-modulated radiotherapy. Charged particle therapy versus photon therapy for paranasal sinus and nasal cavity malignant diseases: a systematic review and meta-analysis. Clinical evidence of variable proton biological effectiveness in pediatric patients treated for ependymoma. Will There Be a Clinically Significant Role for Protons in Patients With Gastrointestinal Malignancies? Proton beam radiation therapy results in significantly reduced toxicity compared with intensity-modulated radiation therapy for head and neck tumors that require ipsilateral radiation. Long-term outcomes after proton beam therapy for sinonasal squamous cell carcinoma. Proton therapy and concomitant capecitabine for non-metastatic unresectable pancreatic adenocarcinoma. Predicted rates of secondary malignancies from proton versus photon radiation therapy for stage I seminoma. Intensity modulated proton therapy versus intensity modulated photon radiation therapy for oropharyngeal cancer: first comparative results of patient-reported outcomes. Long-term survival and toxicity in patients treated with high-dose intensity modulated radiation therapy for localized prostate cancer. Upper gastrointestinal complications associated with gemcitabine concurrent proton radiotherapy for inoperable pancreatic cancer. Long-term single-institute experience with trimodal bladder-preserving therapy with proton beam therapy for muscle-invasive bladder cancer. A dosimetric comparison of proton and photon therapy in unresectable cancers of the head of pancreas. Second cancers among 40,576 testicular cancer patients: focus on long-term survivors. Interfractional variations in the setup of pelvic bony anatomy and soft tissue, and their implications on the delivery of proton therapy for localized prostate cancer. Beam configuration selection for robust intensity-modulated proton therapy in cervical cancer using Pareto front comparison. Proton beam radiotherapy as part of comprehensive regional nodal irradiation for locally advanced breast cancer. Quality of life and patient-reported outcomes following proton radiation therapy: a systematic review. Prospective study of patient-reported symptom burden in patients with non-small-cell lung cancer undergoing proton or photon chemoradiation therapy. A pilot feasibility study of definitive concurrent chemoradiation with pencil beam scanning proton beam in combination with 5-fluorouracil and mitomycin-c for carcinoma of the anal canal. Irradiation with protons for the individualized treatment of patients with locally advanced rectal cancer. Comparative outcomes after definitive chemoradiotherapy using proton beam therapy versus intensity modulated radiation therapy for esophageal cancer: a retrospective, single institutional analysis. Risk of developing second cancer from neutron dose in proton therapy as function of field characteristics, organ, and patient age. Secondary cancers after intensity-modulated radiotherapy, brachytherapy and radical prostatectomy for the treatment of prostate cancer: incidence and cause-specific survival outcomes according to the initial treatment intervention. Four-dimensional computed tomography-based treatment planning for intensity-modulated radiation therapy and proton therapy for distal esophageal cancer. Randomized trial comparing conventional-dose with high-dose conformal radiation therapy in early-stage adenocarcinoma of the prostate: long-term results from Proton Radiation Oncology Group/American College of Radiology 95-09. Conventional external radiotherapy in the management of clivus chordomas with overt residual disease. The role of proton therapy in the treatment of large irradiation volumes: a comparative planning study of pancreatic and biliary tumors. In 1974, Nigro and colleagues from Wayne State reported their experience of 3 patients with anal carcinoma who received neoadjuvant chemoradiation therapy and were found to have a complete response at the time of surgery.
This judgement should only be arrived at following discussion of the options with the patient buy 20mg levitra soft with mastercard erectile dysfunction unable to ejaculate, covering the diagnostic and treatment choices available cheap 20mg levitra soft with amex erectile dysfunction nitric oxide. It is advised discount levitra soft 20mg otc impotence at 33, however purchase 20 mg levitra soft visa erectile dysfunction doctors in ct, that significant departures from the national guideline or any local guidelines derived from it should be fully documented in the patient’s case notes at the time the relevant decision is taken. The grade of recommendation relates to the strength of the supporting evidence on which the evidence is based. B in adult patients who are gCs<15 with indications for a Ct head scan, scanning should include the cervical spine. No evidence was identified to support or refute the safety or efficacy of telephone triage of patients with a suspected head injury. Table 3: Clinical indicators for referral to an emergency ambulance service focal neurological deficit problems understanding, speaking, reading or writing loss of feeling in part of the body problems balancing unilateral weakness any changes in eyesight problems walking. The assessment of amnesia will not be possible in pre-verbal children and is unlikely to be possible in any child aged under five years. The management of individual patients with a head injury, and the formulation and application of guidelines depends upon the use of a widely accepted and applicable method of assessment and classification of the so-called ‘level of consciousness’ as defined by the Glasgow Coma Scale Score. This provides the most useful indication of the initial severity of brain damage and its subsequent changes over time. The glasgow Coma scale provides a framework for describing the state of a patient in terms of three aspects of responsiveness: eye opening, verbal response, and best motor response, each stratified according to increasing impairment. In the first description of the scale for general use, the motor response had only five options, with no demarcation between ‘normal’ and ‘abnormal’ flexion. The distinction between these movements can be difficult to make consistently28,31 and is rarely useful in monitoring an individual patient but is relevant to prognosis and is therefore part of an extended six option scale used to classify severity in groups of patients. This score can provide a useful single figure summary and a basis for systems of classification, but contains less information than a description separately of the three responses. The three responses of the original scale (developed in 1974), not the total score, should therefore be of use in describing, monitoring and exchanging information about individual patients. The guideline development group recommends that the progress of the patient should be recorded on a chart, incorporating the Glasgow Coma Scale and other features. Examination of the cranial nerves, in particular pupil reactivity, and neurological examination of the limbs, focusing on the pattern and power of movement, provide supplementary information about the site and severity of local brain damage. Information about mechanisms of injury, other injuries and complications should also be recorded. Patients with a head injury can be assessed using information from the Glasgow Coma Scale or Score. In view of the widespread use of both systems, the recommendations in this guideline are framed in both terms where appropriate. Annex 3 summarises the procedure for assessing a patient using the Glasgow Coma Scale. Table 5: the Paediatric Coma Scale and Score for use in children under five years of age feature scale score Responses Notation Eye opening Spontaneous 4 To voice 3 To pain 2 None 1 verbal response Orientated/interacts/follows objects/smiles/alert/ 5 coos/babbles words to usual ability Confused/consolable 4 Inappropriate words/moaning 3 Incomprehensible sounds/irritable/inconsolable 2 None 1 Best motor response Obey commands/normal movement 6 Localise pain/withdraw to touch 5 withdraw to pain 4 Flexion to pain 3 Extension to pain 2 None 1 total Coma ‘sCorE’ 3/15 – 15/15 12 4 rEfErral to thE EmErgEnCy dEpartmEnt 4 referral to the emergency department 4. The principal reasons for hospital referral are the existence or potential for brain injury or the presence of a wound that may require surgical repair. Four meta-analyses and six studies either formulated or tested established criteria for predicting intracranial injury. B In addition to the above, children who have sustained a head injury should be referred to hospital if any of the following risk factors apply: clinical suspicion of non-accidental injury ; significant medical comorbidity (eg learning difficulties, autism, metabolic disorders) difficulty making a full assessment not accompanied by a responsible adult social circumstances considered unsuitable. In such cases a specialist paediatrician with responsibility for child protection should be involved. A systematic review of concussion in various contact sports found that the incidence of concussion ranged from 0. This assessment may be undertaken by a practitioner (doctor, or nurse or paramedic with extended training), in a variety of settings, including rural hospitals and surgeries capable of assessing the signs and symptoms detailed in section 4. Arranging transfer of a patient with a head injury to an acute hospital can be a major undertaking because of the distance and/or sea crossings involved. There is evidence to suggest that reduced level of consciousness, loss of consciousness, focal neurology and skull fracture are strong risk factors for requiring surgical intervention in adults and children. If transfer is by air transport this should be to a centre with the resources for undertaking surgical intervention, which will require early notification and discussion with the Scottish Ambulance Service. For patients with other indicators found as a single sign or symptom the clinician will have to use clinical judgement as to the merit of transferring the patient. If transfer is not undertaken appropriate observation of the patient must be put in place. Early imaging, rather than awaiting neurological deterioration, reduces the delay in the detection and treatment of acute traumatic intracranial injury. It may also help to explain the patient’s symptoms and predict a likely pattern of recovery and the need for follow up. These all aim to have as high a sensitivity as possible so few injuries are missed. The assault rate in the Netherlands study (24%) is more similar to Scotland, so the Dutch validation is more generalisable to the Scottish population. The study concluded that there is not one rule that will detect all abnormalities. There are no large prospective studies looking specifically at the risk in anticoagulated patients. C where Ct is unavailable, skull X-ray should be considered in adult patients with minor head injury who do not require transfer for an immediate Ct scan.

